Unique Pharmacological Properties of Cannabidiol
Cannabidiol’s pharmacokinetics are marked by low oral bioavailability (~6%) due to extensive first-pass metabolism. However, its interaction with non-cannabinoid receptors, such as TRP channels and serotonin receptors, sets it apart from other cannabinoids.
Current Clinical Trials involving Cannabidiol APIs
Recent clinical trials have focused on cannabidiol’s potential in treating epilepsy and anxiety disorders. A notable study demonstrated CBD’s efficacy in reducing seizure frequency in patients with Dravet syndrome, a severe form of epilepsy. Another trial is exploring CBD’s effects on anxiety, with preliminary results indicating significant reductions in anxiety levels.
Future Healthcare Potential of Cannabidiol
Cannabidiol’s future in healthcare is bright, with potential applications in treating a variety of conditions, including chronic pain, inflammation, and neurodegenerative diseases. Its non-psychoactive nature makes it suitable for long-term use, and ongoing research continues to uncover new therapeutic avenues. As the understanding of CBD’s mechanisms deepens, it could become a staple in modern medicine, offering relief without the psychoactive effects associated with THC.
Cannabidiol Molecule
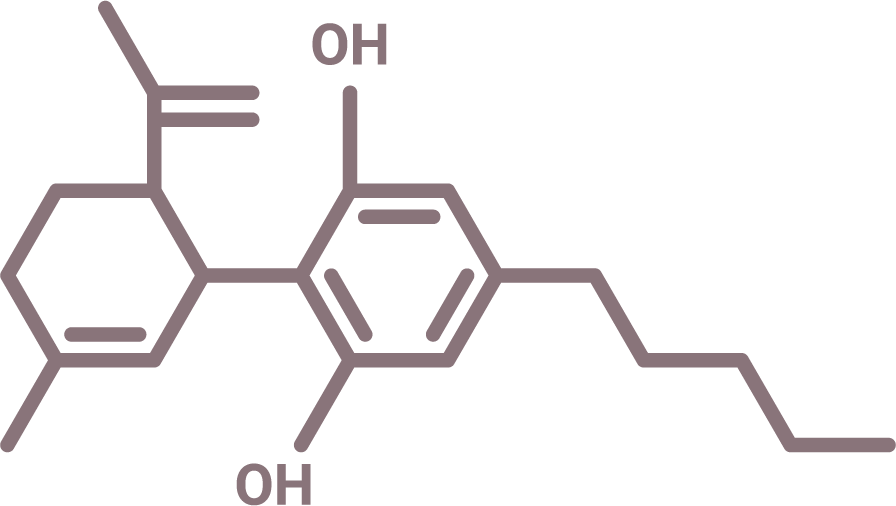
The Distrio Network
The Distrio Network is a content hub for procurement, quality assurance, and technical professionals in the global cannabinoid supply chain. Built using DistrioGPT, with cannabinoid-specific websites delivering blogs, guest stories, and deep dives on the latest research, cannabis-focused professionals will find in-depth and engaging content about these molecules and their markets.
CBD: The Therapeutic Non-Psychoactive Cannabinoid
Cannabidiol (CBD) is a non-psychoactive cannabinoid that has gained popularity for its potential therapeutic benefits. Unlike THC, CBD does not produce a high, making it appealing for medical use. It interacts with various receptors in the body, notably enhancing the activity of serotonin 1A receptors, which contributes to its anxiolytic (anxiety-reducing) effects. CBD’s low oral bioavailability (~6%) is due to its extensive first-pass metabolism. Clinically, CBD has been shown to reduce seizure frequency in epilepsy, particularly in conditions like Dravet syndrome. Ongoing research continues to explore its efficacy in treating anxiety disorders, chronic pain, and inflammation.
CBG: The Emerging Cannabinoid
Cannabigerol (CBG) is a lesser-known, non-psychoactive cannabinoid that is gaining attention for its potential health benefits. CBG is unique because it acts as a precursor to other cannabinoids, including THC and CBD. This means that during the cannabis plant’s growth, CBG is converted into these more prevalent cannabinoids. CBG is being studied for its anti-inflammatory and neuroprotective properties, with early research suggesting it may be beneficial for conditions like inflammatory bowel disease and glaucoma. Although clinical trials are limited, the ongoing research into CBG’s pharmacological effects is promising, highlighting its potential as a therapeutic agent without psychoactive effects.
THC: The Psychoactive Cannabinoid
Tetrahydrocannabinol (THC) is the most well-known cannabinoid, primarily responsible for the psychoactive effects of cannabis. When consumed, THC is rapidly absorbed into the bloodstream, especially when inhaled, with a bioavailability ranging from 10-35%. This means that a significant portion of THC enters the bloodstream and reaches the brain, where it binds to CB1 receptors, producing the characteristic “high.” A unique aspect of THC is its conversion into 11-hydroxy-THC, a metabolite that enhances its psychoactive effects, through the liver. Beyond its recreational use, THC is being studied in clinical trials for its potential to alleviate chronic pain and spasticity associated with multiple sclerosis. These trials aim to harness THC’s analgesic properties while managing its psychoactive effects.
Contact Us